 | |
| Names | |
|---|---|
| Trade names | Daypro, Dayrun, Duraprox, others |
| Other names | Oxaprozinum[1] |
| |
| Clinical data | |
| Drug class | Nonsteroidal anti-inflammatory drug (NSAID)[2] |
| Main uses | Inflammation such as in osteoarthritis and rheumatoid arthritis[2] |
| Side effects | Abdominal pain, stomach ulcers, gastrointestinal bleeding, nausea, poor kidney function, itchiness[2] |
| Pregnancy category |
|
| Routes of use | By mouth |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a693002 |
| Legal | |
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | 95% |
| Protein binding | 99% |
| Metabolism | Liver—65% oxidation and 35% glucuronic acid conjugation. 5% are active phenolic metabolites. |
| Elimination half-life | 54.9 hours |
| Chemical and physical data | |
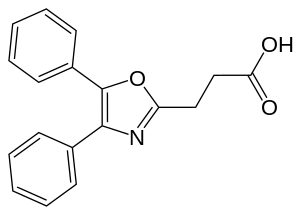
| Formula | C18H15NO3 |
| Molar mass | 293.322 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Oxaprozin, sold under the brand name Daypro among others, is a nonsteroidal anti-inflammatory drug (NSAID), used to treat inflammation in conditions such as osteoarthritis and rheumatoid arthritis.[2] It is taken by mouth.[2]
Common side effects include abdominal pain, stomach ulcers, gastrointestinal bleeding, nausea, poor kidney function, and itchiness.[2] Other side effects may include liver problems, anaphylaxis, heart failure, myocardial infarction, and Stevens-Johnson syndrome.[2] Use in the later part of pregnancy may harm the baby.[2]
Oxaprozin was patented in 1967 and approved for medical use in 1983.[3] In the United States it costs about 2.50 USD per 600 mg dose as of 2021.[2] It is also available in Canada and Japan.[1]
Medical uses[edit | edit source]
In 2015, oxaprozin was one of twenty NSAIDs included in a clinical trial to compare the efficacy of NSAIDs in the short-term treatment of ankylosing spondylitis (AS). The NSAIDs were compared by completing randomized controlled trials of NSAIDs in patients with active AS. Efficacy reported at 2–12 weeks and adverse effects were examined. Efficacy was measured by change in pain score and change in the duration of morning stiffness. A total of 26 trials with a total of 3410 participants were completed (58% of the trials had fewer than 50 participants). While all 20 NSAIDs were found to reduce more pain than the placebo, 15 were found to be significantly better. In regards to the decrease of morning stiffness and the likelihood of adverse events, there was no significant difference between NSAIDs. It was concluded that etoricoxib was more effective in reducing pain of AS, however due to small studies and insufficient evidence, no one NSAID could be determined to be the most effective treatment of AS. After etoricoxib, patients taking oxaprozin experienced the least amount of pain with fewer adverse effects than naproxen.[4]
Dosage[edit | edit source]
The typical dose in adults is 1,200 mg once per day, though doses of 600 mg to 1,800 mg may be used.[2]
Side effects[edit | edit source]
In October 2020, the U.S. Food and Drug Administration (FDA) required the drug label to be updated for all nonsteroidal anti-inflammatory medications to describe the risk of kidney problems in unborn babies that result in low amniotic fluid.[5][6] They recommend avoiding NSAIDs in pregnant women at 20 weeks or later in pregnancy.[5][6]
History[edit | edit source]
Oxaprozin was developed and patented by Wyeth-Ayerst.[7] The US patent 3578671, Oxazoles, was filed November 6, 1967 and published May 11, 1971.[8] Following the filing of the patent, the first description of oxaprozin exhibiting anti-inflammatory properties was outlined in the article Diaryloxazole and diaylthiazolealkanoci acids: two novel series of non-steroidal anti-inflammatory agents. This article was published in Nature in 1968.[9][10] In December 1988, Wyeth-Ayerst licensed the marketing rights for the US, Canada, Puerto Rico, and the Caribbean to Searle.[7]
Daypro became available January 5, 1993. Upon its release, “The Pink Sheet” estimated that the average whole sale price of Searle's Daypro was $112.30 for 100 (600 mg) tablets.[7] The price was comparable to other prescription NSAIDs.
Society and culture[edit | edit source]
Cost[edit | edit source]
The medication has a cost in the U.S. of $41 (USD) for 14 tablets (600 mg)[11]
FDA approval[edit | edit source]
The oxaprozin new drug application (NDA 18-841) was submitted to the FDA on August 10, 1982. The drug was granted an “NDA Day” review on June 15–16, 1992. After Searle agreed to complete seven Phase IV postmarketing studies on October 22, the FDA approved Daypro on October 29, 1992.[7]
Since the approval of Daypro by Searle, other companies have submitted abbreviated new drug applications (ANDAs) to the FDA. Daypro by Searle is listed as the Reference Listed Drug to prove the bioequivalence of the ANDAs. Below is a table listing all of the approved oxaprozin products.
| Company[12] | FDA Approval Date[12] |
|---|---|
| GD Searle | Oct 29, 1992 |
| Apotex Inc | Sep 2, 2004 |
| Dr. Reddy's Labs LTD | Jan 31, 2001 |
| Ivax Sub Teva | May 13, 2002 |
| Sandoz | Jan 31, 2002 |
| Sun Pharm Inds Inc | Jan 3, 2002 |
| Teva | Jul 3, 2002 |
Recalls[edit | edit source]
Advantage Dose LLC recalled oxaprozin tablets on November 26, 2008. The company was not in conformance with cGMP. (Recall #D-837-2009)[13]
References[edit | edit source]
- ↑ 1.0 1.1 Society, Swiss Pharmaceutical (2000). Index Nominum 2000: International Drug Directory. Taylor & Francis. p. 768. ISBN 978-3-88763-075-1. Archived from the original on 10 November 2021. Retrieved 10 November 2021.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 2.8 2.9 "Oxaprozin Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 18 January 2021. Retrieved 10 November 2021.
- ↑ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 520. ISBN 9783527607495. Archived from the original on 2020-08-06. Retrieved 2020-10-17.
- ↑ Wang, Runsheng (6 Aug 2015). "Comparative efficacy of non-steroidal anti-inflammatory drugs in ankylosing spondylitis: a Bayesian network meta-analysis of clinical trials". Annals of the Rheumatic Diseases. 75 (6): 1152–1160. doi:10.1136/annrheumdis-2015-207677. PMID 26248636. S2CID 20375113. Archived from the original on 25 January 2020. Retrieved 29 June 2019.
- ↑ 5.0 5.1 "FDA Warns that Using a Type of Pain and Fever Medication in Second Half of Pregnancy Could Lead to Complications". U.S. Food and Drug Administration (FDA) (Press release). 15 October 2020. Archived from the original on 16 October 2020. Retrieved 15 October 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ 6.0 6.1 "NSAIDs may cause rare kidney problems in unborn babies". U.S. Food and Drug Administration. 21 July 2017. Archived from the original on 17 October 2020. Retrieved 15 October 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ 7.0 7.1 7.2 7.3 The NDA Pipeline 1992. Chevy Chase, MD: F-D-C Reports, Inc. 1992. pp. I-462.
- ↑ Oxazoles, archived from the original on 2016-05-06, retrieved 2015-12-07
- ↑ Brown, K. (July 13, 1968). "Diaryloxazole and Diarylthiazolealkanoic Acids: Two Novel Series of Non-steroidal Anti-inflammatory Agents". Nature. 219 (5150): 164. Bibcode:1968Natur.219..164B. doi:10.1038/219164a0. PMID 5301713. S2CID 4214027.
- ↑ The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals. Whitehouse Station, NJ: Merck Research Laboratories. 2001.
- ↑ "Oxaprozin Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 18 January 2021. Retrieved 5 April 2021.
- ↑ 12.0 12.1 "Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations". www.accessdata.fda.gov. Archived from the original on 2016-08-17. Retrieved 2015-12-02.
- ↑ "FDA Enforcement Report" (PDF). FDA.gov. June 24, 2009. Archived (PDF) from the original on February 10, 2017. Retrieved Dec 2, 2015.
External links[edit | edit source]
| Identifiers: |
|
|---|
.svg.png)
.svg.png)